Biochemistry of photosynthesis
Biochemistry of photosynthesis
We understand much of the basic mechanism of the light reactions of photosynthesis by which two photosystems use light energy to drive electrons through an electron transfer chain. This is the so-called Z-scheme of Hill and Bendall, proposed over 60 years ago. Water is oxidised, with oxygen as a waste product, and electrons are available – typically for CO2 fixation.
However, there are many aspects and proteins involved that we still don’t fully understand. One of these is a curious cytochrome, called cytochrome c6A, that we discovered twenty years ago.
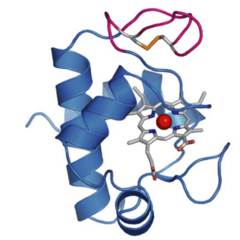
Figure 1: Structure of cytochrome c6A. Note the heme group, and the (magenta) extended loop containing two cysteines (yellow). From Marcaida et al. (2006) Journal of Molecular Biology doi: 10.1016/j.jmb.2006.05.065.This cytochrome is similar to the better-known cytochrome c6, which can transfer electrons from the cytochrome b6f complex to Photosystem I. However, we showed that cytochrome c6A has too low a redox midpoint potential to carry out that function (Wastl et al. (2003) Nature https://doi.org/10.1038/424033b). It also has a cysteine-containing insertion in a loop region of the protein. Its function remains unknown. In spite of that, the protein is widespread and well conserved among photosynthetic organisms (Slater, Kosmützky et al. (2021) Genome Biology & Evolution doi:10.1093/gbe/evab146) and we are keen to understand its function.
More information:
“The evolution of the cytochrome c6 family of photosynthetic electron transfer proteins” *Slater B, *Kosmützky D, Nisbet RER, Howe CJ (2021) Genome Biology & Evolution, 13:8 evab146, doi:10.1093/gbe/evab146
"The novel cytochrome c6 of chloroplasts - a case of evolutionary bricolage?" Howe CJ, Schlarb-Ridley BG, Wastl J, Purton S, Bendall DS (2006) Journal of Experimental Botany 57:13-22, doi: 10.1093/jxb/erj023.